When an element gains an electron, we can think of it as it is being. 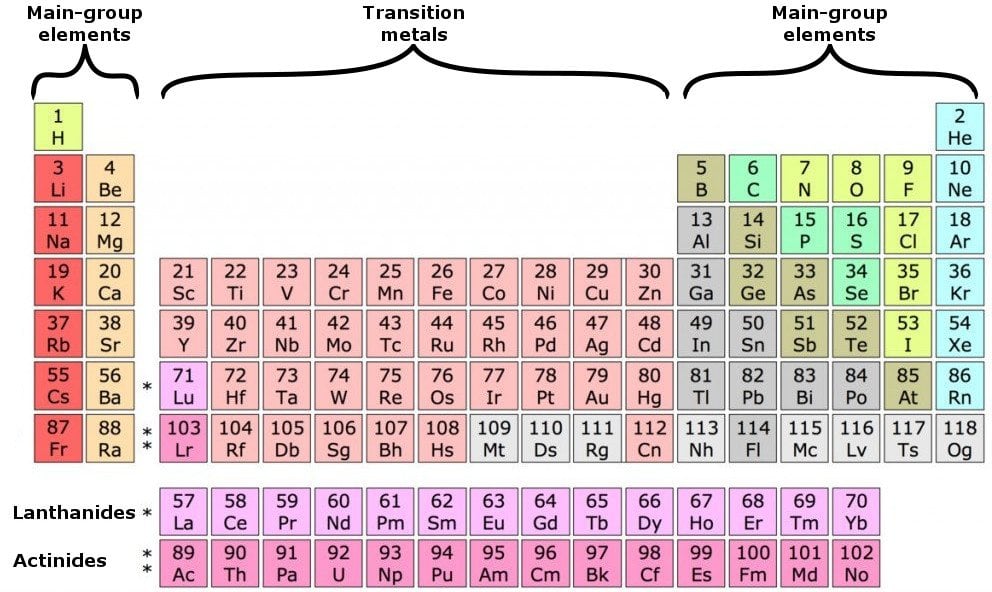
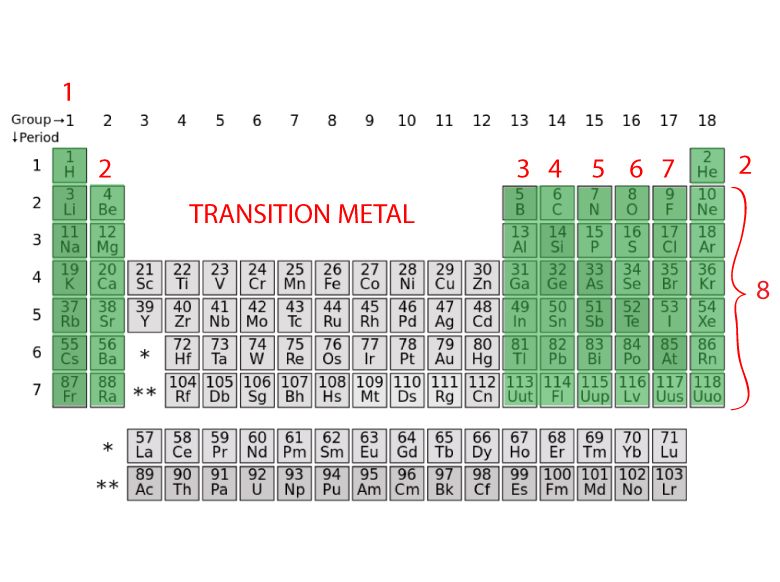
The valence electrons are also responsible for determining the electrical conductivity nature of an element. Each row in the periodic table is called a period. coordinate covalent bonds (also called dative covalent bonds).This is known as chemical bonding and serves to build atoms into molecules or ionic compounds. By moving electrons, the two atoms become linked. Another form of ionic bonding involves an atom giving some of its electrons to another atom this also works because it can end up with a full valence by giving up its entire outer shell. This can be achieved one of two ways: an atom can either share electrons with neighboring atoms, a covalent bond, or it can remove electrons from other atoms, an ionic bond. Group 1 alkali metals are therefore very reactive, with lithium, sodium, and potassium being the most reactive of all metals.Įvery atom is much more stable, or less reactive, with a full valence shell. As a general rule, the fewer electrons in an atom's valence shell, the more reactive it is. The periodic table shown above demonstrates how the configuration of each element was aligned so that the last orbital filled is the same except for the shell. 
Therefore, elements with the same number of valence electrons are grouped together in the periodic table of the elements. The number of electrons in an atom's outermost valence shell governs its bonding behavior. ** Except for helium, which has only two electrons. * Valence electrons are not generally useful for transition metals. With the exception of groups 3–12 ( transition metals), the number within the unit's place identifies how many valence electrons are contained within the elements listed under that particular column. The number of valence electrons of an element is determined by its periodic table group (vertical column) in which the sup element is categorized. We can see from the electron configuration of a carbon atom1 s2 2 s2 2 p2 that it has 4 valence electrons (2 s2 2 p2) and 2 core electrons (1 s2 ). Helium is a member of the noble gases and contains Valence shell electrons (or, more simply, the valence electrons) are the electrons in the highest-numbered shell, or valence shell, while core electrons are the electrons in lower-numbered shells. This helium (He) model displays two valence electrons While inner electrons (those not in the valence shell). Safe Weighing Range Ensures Accurate Results The number of valence electrons Valence Electrons Valence electrons are the highest energy electrons in an atom and are therefore the most reactive. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
